Note: Mention of trade names and growth-promoting implant manufacturers in this publication is necessary as FDA approvals are specific to trade names and manufacturers.
The US Food and Drug Administration (FDA) is updating information to beef cattle producers concerning a group of growth-promoting implant products (implants) that do not specify on the labeling whether reimplantation is approved. Three important developments must be considered by all cattle producers:
- A document (letter) released on December 10, 2021 (FDA Letter to Industry on Beef Cattle Ear Implants | FDA) provides the basis for implant labeling clarification to the industry.
- In this document, the FDA set June 2023 as the target date for the sponsors of the group of implant products without specified labeling on reimplantation to begin introducing updated labeling regarding reimplantation within a production phase. (Production phases are outlined in graphics and tables below)
- In August of 2022, specific products manufactured by Zoetis received approval from FDA on expanded labels for beef implant products (Zoetis Receives Expanded Labels for Beef Implant Products | Zoetis US).
Production phases, as defined in the Cattle Feeding Variants and Variant Subsets (from CVM GFI #191 Changes to Approved NADAs - New NADAs vs. Category II Supplemental NADAs | FDA), are the boundaries within which a specific implant product is approved for. From FDA Letter to Industry on Beef Cattle Ear Implants: It is important to note that reimplantation across production phases is not affected by these pending label changes. Beef cattle are and will still be allowed to receive multiple implants across different phases of production when using implants approved for that specific phase. Cattle can also receive more than one implant within a production phase, but only if that implant is approved for repeated administration. Figure 1 below depicts the general growth sequence of a given calf from birth to harvest. Figure 2 below depicts production phases as described in the Cattle Feeding Variants and Variant Subsets (from FDA GFI 191; CVM Guidance for Industry #191).
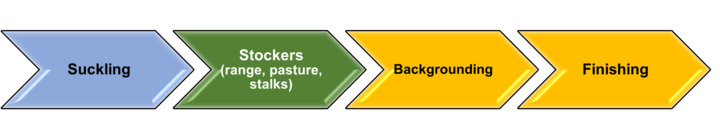
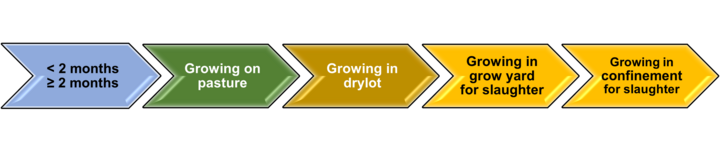
Colors in Figures 1 and 2 are meant to represent matching phases between the ones occurring in the industry and those described by FDA GFI 191; CVM Guidance for Industry #191. Within the calfhood phase (suckling), FDA recognizes two subsets: “Less than 2 months of age”, and “2 months of age and older”. Also, FDA recognizes a “Growing in drylot” phase; but currently, there are no growth-promotion implants approved for this phase. Therefore, interpreting reimplanting under FDA production phases leads to group “Backgrounding” in Figure 1 under “Growing in grow yard for slaughter” in Figure 2 (this is emphasized by matching color background in Figures 1 and 2), which is considered a single phase of production under: “Growing in confinement for slaughter”.
The reader is encouraged to review and understand the similarities and differences between depictions of production phases in Figures 1 and 2. Items to note:
- Colors in Figures 1 and 2 are meant to represent matching phases between the ones common in the cattle industry and those described by FDA GFI 191; CVM Guidance for Industry #191.
- FDA recognizes two subsets for suckling calves: “Less than 2 months of age”, and “2 months of age and older” (Synovex® C is the only implant approved for calves 45 days or older; Component® E-C and Synovex® One Grower are approved for calves 2 months of age or older)
- FDA recognizes a “Growing in drylot” phase but there are no growth-promotion implants approved for this phase.
- FDA recognizes only one phase of production encompassing backgrounding and finishing.
- Effectively, a backgrounding phase, as known to the cattle industry, fits only under “Growing in grow yard for slaughter”. Therefore, cattle under this production phase are considered by FDA as cattle “Growing in confinement for slaughter”.
- Therefore, unless product application sequence (reimplantation) is approved by FDA after June 2023, no reimplantation is permitted within this combined production phase.
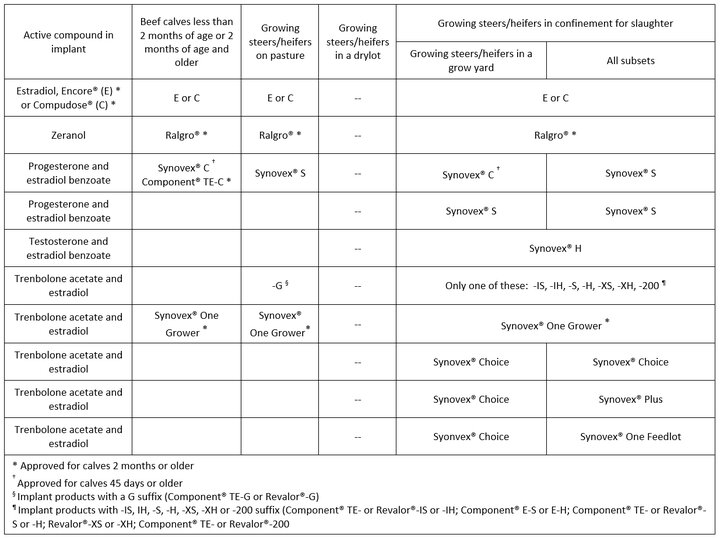
Table 1 below was conceived with the following thoughts in mind:
- Each production phase is depicted according to FDA GFI 191; CVM Guidance for Industry #191 and using the basic hormonal compound each product is comprised of.
- Commercial products listed within each production phase are intended to represent single not sequential implant application (reimplantation) unless clearly specified within the footnotes.
- The phase: “Growing steers/heifers in a drylot” has no specific implant products approved for it but was left in the Table to emphasize this observation.
- Therefore, using two implant products in sequence (reimplanting) for cattle fed in confinement for slaughter “in group pens and fed a progressively high-energy diet ad libitum as their sole ration until slaughter” (backgrounding or finishing) will only be on label if implant products selected are approved for reimplantation.
- For a given production phase, please review the entire column for product choices approved for single or sequential application (reimplantation) within that phase.
- Concurrently, not listing a product under a given production phase (blank cells) does not mean that products that are approved for that production phase do not exist (see note 5 above).
- Lastly, products listed across production phases on the same row are not intended to be construed as implanting strategies across production phases (listing these products within a given cell was for convenience in saving space).
Producers are encouraged to visit with professionals who sell and support the sale of implants to better understand what situations will lead to on or off-label use of implants starting June 1 of 2023. Producers are also welcome to reach me at the Cuming County Extension office regarding a specific situation your operation may be considering.
Interviews with the authors of BeefWatch newsletter articles become available throughout the month of publication and are accessible at https://go.unl.edu/podcast. You can subscribe to the BeefWatch newsletter here: http://go.unl.edu/Beefwatch_subscribe.
Topics covered:
Backgrounding & feedlot, Backgrounding/stocker/yearling, Implants

